Are you ready to kick your oil infusion skills up a notch? An alcohol intermediary oil infusion harnessing the solvent powers of alcohol might sound a bit intimidating, but it’s surprisingly simple to do.
Before we get to the hows of making an alcohol intermediary oil infusion, let’s take a quick look into the whys.
First, some essential resources
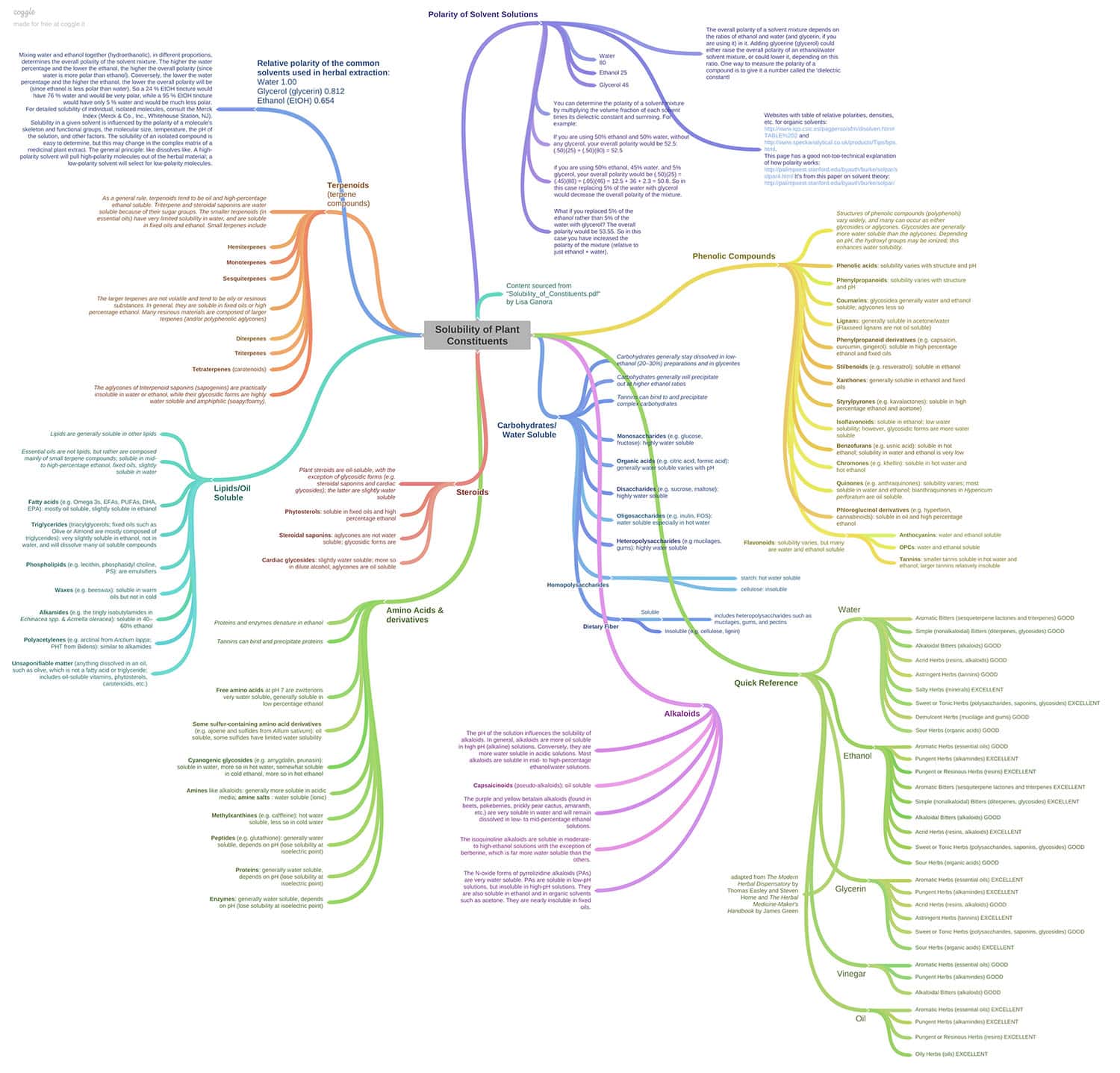
Creating an effective herbal infusion depends on matching your herb’s constituents with their preferred solvent(s). In chemistry, like dissolves like, so it’s useful to know a little about solvent polarity and the corresponding polarity of different plant constituents. But when you’re working with a whole herb, you’ve got something a lot more complex and synergistic than a simple isolated constituent, so these concepts become generalities rather than hard-fast rules.
Fortunately, there’s a whole lotta of resources out there that can help you to figure out which solvents are most effective at extracting what constituent — without revisiting your sophomore year chemistry class.
It’s essential to have trusted source (or two) for determining the specific phytochemicals in the herbs you’re working with. I bounce back and forth between David Hoffman’s Medical Herbalism: The Science Principles and Practices Of Herbal Medicine and Kerry Bone’s Principles and Practice of Phytotherapy: Modern Herbal Medicine. Both books contain extensive, detailed herbal monographs, including a list of primary constituents for each herb. If I can’t find an herb in one book, I’ll almost certainly find it in the other.

Now that we know the primary phytochemicals in our herb, which solvent(s) are best to pull them out? Richo Cech has a helpful section in chapter 5 of his book Making Plant Medicine, “Solubility Factors” that covers some major categories of constituents such as alkaloids, volatile (essential) oils, polysaccharides, resins, etc., examples of herbs that contain them, and what solvents will best extract these constituents. There’s also an extensive chapter on Solvents and which constituents they affect in James Green’s The Herbal Medicine Maker’s Handbook.
For the more advanced herbalist, if you really want to geek out on the chemistry of herbal constituents, Lisa Ganora’s Herbal Constituents: Foundations of Phytochemistry is a goldmine of information. While it’s a good mix of conversational and technical writing, be warned that this is a chemistry text book and is definitely not light reading.
For even more happy geek time, check out this fabulous solubility flowchart that illustrates what solvents extract which plant constituents, adapted from Lisa Ganora’s book and The Modern Herbal Dispensatory by Thomas Easley and Steven Horne and The Herbal Medicine-Maker’s Handbook by James Green.
I just wish I knew who created the flowchart. If anyone knows, please let me know in the comments so I can give them credit!
Solvents & solubility
When you infuse an herb in oil, it will draw out some the oil-soluble bits such as gums and resins, some of the volatile oils, certain oil-soluble vitamins (A, D, E, K), plant sterols and terpenes (oil-soluble carotenoids give plants their orange or yellow pigment).
A vibrant red (fresh) St. John’s wort-infused jojoba oil:

Compared to alcohol, water and vinegar, though, oil is a relatively weak solvent. Adding a bit of alcohol to the process will free more of a plant’s alkaloids, resins, flavonoids, glycosides, terpenes and volatile oils, camphors and vitamins. Alcohol also tends to do a better job of capturing certain plant pigments. Chlorophyll, for example, is captured quite well by ethyl alcohol, giving your infused oil a more vibrant color vs. one made without the alcohol intermediary step.
Alcohol intermediary
So to draw out a more complete array of constituents in an oil maceration, ethyl alcohol can be used to do a quick pre-infusion before covering the herbs in oil. This added step draws out the alcohol soluble constituents in addition to the oil soluble ones, effectively increasing your infused oil’s potency.
Simply place your dried herbs in the vessel you’re going to use for the oil infusion. If the herb is in larger chunks, grind or crush into smaller bits using a coffee grinder or mortar and pestle. Barely moisten the herbs with a shallow layer of high-proof grain alcohol (the highest you can find, preferably 190 proof *). Stay away from isopropyl alcohol, though — the smell, even after maceration, will be overpowering.
Pro tip: An easy way to evenly moisten the herbs without drenching them is to place your alcohol in a clean spray bottle and mist the herbs until barely moist.

Cover the jar with a tight-fitting lid and allow the herbs to macerate in the alcohol overnight. During this stage, the goal is extraction, not evaporation, so limiting airflow helps preserve the alcohol’s solvent strength and reduces oxygen exposure.
In the morning, and you can proceed with a normal oil infusion. The alcohol intermediary method benefits from a heated oil infusion method, because the heat will ensure that the alcohol and water evaporates out. This is particularly true if you used a lower proof alcohol. 151-proof Everclear is about 75% alcohol, which means the remaining 25% is water, and you really don’t want water in your final infused oil.
Remember to leave the lid off or vented during the heated infusion stage to allow for evaporation of any alcohol and moisture remaining in the oil.

In addition to the added potency, an alcohol intermediary infusion done correctly should also improve your infused oil’s shelf life. There should be no appreciable alcohol content left in the oil once the oil maceration is complete, but the extra alcohol step reduces the microbes introduced into the oil.
Remember to label your jar with the herb, type of oil used and date (a note about it being an intermediary infusion might also be useful) and store in a dark, climate controlled location. It should last well beyond the estimated 2-year lifespan of a regular dried herb-infused oil.
* We can’t buy 190-proof anything here in Idaho, but I’m fortunate that we live within a pretty easy drive to the Washington border, where 190-proof spirits are a thing. Each time I pick up a few bottles of 190-proof Everclear, I always feel compelled to make sure the checkout clerk understands that I’m NOT drinking it, I swear! I’m not that kind of an alcoholic thankyouverymuch… I’m TINCTURING! ☺
Frequently asked questions
Short answer: no.
When used correctly (a very small amount, brief contact), high-proof ethanol does not damage plant constituents. Ethanol is actually one of the gentlest and most compatible solvents in herbal medicine. It dissolves and mobilizes constituents — it doesn’t destroy them.
Concerns about alcohol “killing” herbal properties usually stem from confusion with excessive heat, oxidation, or prolonged soaking — not from alcohol itself.
There aren’t really problem plants — but there can be problem goals.
If your primary intention is to preserve large amounts of:
- mucilage (e.g., marshmallow root),
- long-chain polysaccharides,
- or starches,
then alcohol wouldn’t be the ideal primary solvent. That said, a brief alcohol intermediary step does not destroy these constituents — it simply doesn’t preferentially extract them. They remain available to the oil phase of the infusion.
Not for the alcohol intermediary step.
Flowers, leaves, roots, bark, seeds — all can be lightly moistened with the same high-proof alcohol. The key variable here is amount, not proof. You want the herbs barely damp, not saturated.
Yes! For dried herbs, one high-proof alcohol (190-proof if available, or the highest proof you can reasonably obtain) will work across the board. You don’t need different proofs for different herbs or plant parts in this method.
Fresh herbs can be used, but they require more care. Because fresh plants already contain water:
- Use alcohol sparingly
- Ensure thorough evaporation during the heated oil infusion stage
- Consider an airtight cover during the alcohol step to reduce oxidation
The same high-proof alcohol is still appropriate — the difference is moisture management, not solvent choice.
Not if the infusion is done correctly.
A heated oil infusion with the lid off or vented allows alcohol and water to fully evaporate. There should be no appreciable alcohol left in the finished oil.
When done properly, the alcohol intermediary step reduces microbial load, and the heated infusion drives off residual moisture. Together, these factors often extend shelf life beyond that of a standard dried-herb oil infusion.



32 Responses
Thank you for this!! I was wondering if you leave the cover off or on during the heated oil infusion? My plan was to place the in my dehydrator at a temp between 100-105 F and wasn’t sure. Thanks I’m advance for any further imparted knowledge🌻
Hi Stefanie! Yes, when doing a heated oil infusion, definitely leave the cover off (or at least vented) to allow evaporation of any remaining moisture in the alcohol or herbs. Your intuition is spot on! Using a dehydrator is a great idea too. 100-105 F is a good range for preserving the quality of the herbs and preventing them from overheating or burning.
I’m looking at making a marijuana infused oil using this method but only have access to 40% alcohol. Question 1… do I still need to decarb the herbs and question 2. Will there be too muvh water from the 40% alcohol or will I manage to burn this off if I heat it in a slow cooker or rice cooler. I’m worried the water content may reduce the shelf life of my balms. I’d appreciate any suggestions. Thanks
Hi Linda! I don’t have any direct experience working with THC (since it’s not legal in my state) but my understanding is that you can decarb the herb at the same time it’s infusing in oil – the decarboxylation process will just take a bit longer, since oil acts as a buffer and can slow down the conversion of THCA to THC. Ideally, you’d want to use a high-proof alcohol, such as Everclear for your intermediary oil infusion to better extract the resinous compounds and minimize the water content in your balm, but you may be able to get away with using 40% alcohol if you heat the oil infusion low and slow for a longer period of time to evaporate the water content (which you would be doing anyway for the decarbing process). Be sure to leave the lid off your slow cooker to allow the water to escape. Good luck!
I have the same problem as above, access to approx 75% alcohol
I fail to logically see how water can evaporate at such a low temperature. Can’t water only evaporate at 100 degrees Celsius?
Will the water actually evaporate slow boiling at lower temps?
Thanks
Davy
Hi Davy! While it’s true that water’s boiling point is 100C (212F), evaporation can occur at lower temperatures. Water molecules can escape from the liquid phase and enter the air as vapor at room temperature, although the rate of evaporation will be much slower than when heat is applied. Low heat (that won’t denature your herb) will speed up this process.
Hello, i did this method with calendula/tallow but i believe i added too much alcohol. the water bath went 12 hours and was at 150 degrees at its highest but i can still smell alcohol pretty strong in the oil. Should I leave it alone and continue the process or should i try to increase heat to try to burn off the remaining alcohol. I’m not sure how high i can go with heat in water bath before it will damage the medicinal properties of the plant. Thanks so much and I love your site!!
Hey Lisa, Thanks so much for the kind words! I would leave the temperature on the lower side (you really don’t want the oil itself to heat much beyond 110-115 or so) and continue your water bath infusion until all of the alcohol has evaporated. (Good job letting you nose be your guide to determine when the infusion is actually “done”!)
Richo Cech recommends that fresh herbs be infused for a full week and 2 weeks for dried herbs, so your infusion may benefit from a longer maceration time to evaporate the remaining alcohol. If you used a lower proof alcohol (like 151 Everclear) it could take a bit longer. Good luck ♥
Hi There,
I have been following the instructions from Mountain Rose Herbs on doing an alcohol intermediary oil infusion method using a blender. You soak the alcohol and herbs overnight and then the next day blend them in oil for 5 minutes using a blender-the oil gets warm. I was wondering if you had done this before and what your success rate was. I have a dehydrator and could easily use that after the overnight soak. How long do you recommend dehydrating the herb/oil mixture (top off)? I really was looking to see if one way was better than the other or maybe if it was just the same. Thanks in advance for your help. I am appreciative of the information.
Libby
Hi Libby! The blender method works extremely well (you can read my version of this process here: https://redheadedherbalist.com/a-simple-way-to-supercharge-your-tinctures-oil-infusions/ A Simple Way to Supercharge Your Tinctures & Oil Infusions).
Most of the details are similar, though I would add that using a very high proof alcohol is important – 151 minimum and 190 if you have access to it. It’s so much easier to get the water out if there’s not a ton there to begin with. Good thinking about following up with the dehydrator. If I used a lower proof alcohol and/or fresh plant material, I’ll transfer the macerated oil to a jar inside my dehydrator cabinet, and hold it (uncovered) at ~110F for another day or two, just to be certain all of the moisture is evaporated out of the oil. The dehydrator step doesn’t hurt anything and it gives you peace of mind that the moisture is completely gone. I’ve been using this method (even with fresh plant material) for about a year now, and haven’t had any oils go moldy.
I hope this helps! Please let me know how yours turns out! ♥
Hello! Thank you for this great blog post! I love the amount of details that you provide and the books you mentioned. I would like to try the Alcohol Intermediary Herb-infused Oil method you describe on this blog post. I plan to use the herb-infused oils for skincare products for the face and I want to ensure that no alcohol or water remains in the oil infusion. Other than the smell test, is there anything else I could to to test if water or alcohol remain after 2 weeks of infusing the oil at low heat?
Thank you very much!
Che
Thank you so much for the kind words, Che! I have never tried to measure residual alcohol content in my infused oils. I always use 190-proof, so there’s minimal water to deal with. If you’re concerned, you might try letting the alcohol part of the alcohol intermediary infusion sit a bit longer before adding it to the oil. 2 weeks at low heat should be more than enough time to allow any residual water content to evaporate out of your oil. As far as measuring goes, I don’t have any direct experience with these tools, but you might have luck with a specialty hydrometer designed for oil products such as this one. Good luck, and if you find an accessible solution, please report back!
Thank you very much, Redheaded Herbalist (btw, I love your name!)! This information is very helpful. I will let you know if I find any cost-effective and practical way to test for any residual alcohol content. In California I can only get 75.5% alcohol. Would letting the alcohol intermediary infusion sit 3-4 days be too long? If I let it sit for too long, would I lose the properties extracted by the alcohol? (can they evaporate?)
Thank you very much!!
It depends on the herb and the constituents you’re trying to extract. You probably would risk losing *some* of the herb’s volatile constituents (like terpenes or phenolic compounds) the longer it sits. And, unfortunately, measures that minimize the loss of volatile compounds would also slow down the evaporation of water from the alcohol. Of course, if you’re primarily interested in capturing something like resin from the herb, then evaporation is less of a concern.
Thank you very much!! Since higher proof alcohol is not sold in my state, I believe another option for me would be to use undenatured alcohol. Do you have any experience with that?
Also, could the intermediary method be used with glycerin instead of oils?
Thank you very much!
I just made an infusion of lavender and oil using my ne Hello Hi device. I think it’s intended use I’d to infuse marijuana. However, I used it for my herbs and used the alcohol method. I think I put too much in the formulation. What can I do. It does smell of alcohol. The machine has a de carb setts which I will later use instead of the alcohol to determine the difference. In the meantime do I throughout or try to salvage my infuse and how much of my valuable properties have I lost. I was making for my grandson who suffers from eczema. Please help a new herbal beginner. Thank you!
Hi Eretta, I would allow the infusion to sit uncovered for a bit longer to allow the alcohol more time to evaporate. It might take a while, but alcohol is volatile, so it will evaporate eventually. You can try holding the infusion at a low temperature (also uncovered) to speed up the evaporation process. Avoid overheating it, though, since the volatile aromatic compounds in lavender will also evaporate. Keep it under 110F, and see if the scent improves. Good luck! ♥
I am trying the alcohol method and using 91% isopropyl since that’s the best I have. I let it sit in a jar (with a plastic lid on it) overnight thinking that the alcohol was not supposed to evaporate until it had done its work. I have two questions.
1. Should I take the lid off and just cover it with cheesecloth so more alcohol can evaporate BEFORE I add in the oil? Is there any reason that this would be bad for the end product?
2. Is the BLENDER step necessary, or can I simply stir in the oil and then do the warming process? When using the vitamix, it is impossible to get everything out so you lose a lot of the oil. Versus just adding the oil to the jar the herbs are already in. Does that make sense? I already crushed my herbs (comfrey) really well before adding the alcohol. While I’m sure that blending in the vitamix would break them down more, it seems like it would also make it next to impossible to strain all plant stuff out without using a lot o absorbent filter material which will also reduce the amount of oil for the finished product.
Hi Nancy! Since our goal is to have an infused oil with no remaining water OR alcohol left in it, you do want to allow for evaporation. I usually cover with cheesecloth and a rubber band during the alcohol stage, but I did have one person report browning/oxidation happening to her comfrey when she did the alcohol maceration, so it might not be a bad idea to use an airtight cover during that step to reduce oxygen exposure (and the risk of oxidation). In order to get the rest of the alcohol/water out of your preparation, though, you’ll need to apply gentle heat (no more than 100F) for a day or two to encourage the remaining water/alcohol to evaporate out of the oil.
The blender step is not at all necessary – it simply speeds up the process. You can make a perfectly good alcohol intermediary oil infusion using any of the methods described here: https://redheadedherbalist.com/making-herbal-oil-infusions/
Thanks for the response! I ended up not blending it. And I left it out for ages to try to evaporate more alcohol. I did heat in a double boiler, not boiling though, just to warm and help evaporate. And it took ages to strain, so it sat open while straining. I didn’t end up having time to make salve yet, so I just put the oil in the fridge. It still smelled a bit of alcohol, but I decided not to worry about it. It is SO bright/intense colored, which is really fun.
I’m wondering if there is a bare minimum time to keep dry herbs in the alcohol. I was wanting to do an oil infusion ready tonight with a magical butter maker. So just like a 3 hour heat/infuse time. But I just mixed with the alcohol then thought oh, how short of time can I do with the alcohol…. Any suggestions? I may have to wait overnight but wondered…🤪 Margaret
Hi Margaret! It doesn’t have to be overnight. If you’re in a rush, even 30 minutes of an alcohol pre-treatment can help to break down plant cell walls and begin extracting medicinal compounds. And since you’re using the MagicalButter Machine, the heat should take care of any residual alcohol.
Thank you for writing this. Appreciate you sharing your knowledge. If you sell the herbal infusion by itself, or use it in salves, balms etc., do you add ethyl alcohol in the ingredients list?
No, ethyl alcohol does not need to be listed in the ingredient list if it is not present in the finished product. In a properly done alcohol-intermediary oil infusion, the alcohol is fully evaporated during the heated oil stage, so it functions as a processing aid, not a final ingredient.
I don’t have a dehydrator. Can I leave the alcohol treated herbs in the oven instead of a water heat bath? What temp is best? Thank you
You don’t necessarily need a dehydrator, it’s just what I like to use. Water baths (jar-in-water method) using a slow cooker or Instant Pot, with the jar elevated on a trivet or canning rings and the lid vented work well. My article Making Herbal Oil Infusions goes into more detail: https://redheadedherbalist.com/making-herbal-oil-infusions/
In most cases, a regular oven is not a good choice, because even the lowest setting on many ovens runs far hotter than we want for herbal oils. For hot oil infusions (including alcohol-intermediary oils), you’re aiming for warm, not hot – ideally 95–105F. Temperatures above that can drive off volatile compounds and damage the medicinal properties of the plant.
Hello! So I would like to start using the alcohol intermediary method for my oil infusions but I’m a bit confused. Can I use everclear for all infusions or should I be using different proofs depending on the makeup ie: flower, leaf, root etc.? I only use dried material. Thanks in advance!
Hi Vanessa, Great question and a common point of confusion. High-proof Everclear is appropriate for all alcohol-intermediary oil infusions, regardless of whether you’re working with fresh or dried flowers, leaves, roots or bark. In the alcohol-intermediary method, the alcohol is only doing a brief, shallow pre-extraction. You’re not making a tincture or trying to dial in an exact alcohol percentage for selective extraction, you’re simply using alcohol’s strong solvent power to loosen and free constituents that oil alone doesn’t grab well. High-proof ethanol works best for this because it extracts a broad spectrum of alcohol-soluble constituents (alkaloids, flavonoids, glycosides, resins, volatile oils, pigments, etc.) while introducing minimal water (which is especially important when your end goal is an oil) and evaporates cleanly and completely during the heated oil infusion stage.
Hello, thank you for this write up; I will be bookmarking your site! I would love if you had the time to weigh in on a question I have. I made a massive batch of an AIIO blend using 95% cane and the Ethyl Oil Salve method from Green’s THMM Handbook. I am currently double boiling another third of the batch of the strained oil, but what I’ve poured off already still smells very strongly of the alcohol. I did an hour and a half and it seemed to be gone, but after it cooled off a bit and settled in the jar I was able to smell it. Would you say that it just needs more time, and if so how long? I have an induction stove which seems to heat inconsistently, and am really trying to avoid boiling my oil! Also, what are your thoughts on keeping the ethyl oil as-is (for a few months or even years), then double boiling it much later? Any insight would be appreciated!
Hi K, thanks for the sweet message! As you’re finding, large batches — especially on an induction stove that cycles heat — often need more time to fully evaporate the alcohol. Recommended infusion times vary widely among herbalists: a day or two is common; The Herbal Academy suggests 4–8 hours (or longer), while Richo Cech recommends about a week for fresh herbs and two weeks for dried.
Gentle, extended heat with airflow is key. Keep the oil uncovered or loosely vented, stir occasionally, and hold it warm (not hot or bubbling). I’m personally cautious about double boiling my oil infusions since steam or condensation can reintroduce moisture, though many herbalists do recommend it.
And since you used 95% ethanol, water content is minimal and likely already evaporated, so storing the oil as-is for a period of time is likely safe. Ethanol doesn’t bind to oil, and you can return to it months later and use gentle heat to drive off any remaining alcohol.
Hope this helps! ♥
Thank you so so much; I appreciate your comprehensive reply! Happy February!
Hi! Thank you for the helpful article. Do you know if the alcohol intermediary method can be used for heat sensitive oils like cacay, hemp or rosehip? If so, would you omit the heating step and only blend? Thanks in advance!